AGR4301 Advanced Crop Physiology Laboratory Manual
- PlantHouse Enterprise
- Mar 31
- 6 min read
Download Easy Leaf Area app here: https://play.google.com/store/apps/details?id=com.eclipsim.gpsstatus2&hl=en
Download GPS Status & Toolbox app here: https://play.google.com/store/apps/details?id=com.eclipsim.gpsstatus2&hl=en
1. INTRODUCTION
Duckweed (Lemna spp.) is a fast-growing aquatic plant widely used as a model organism in plant physiology due to its rapid vegetative reproduction, simple structure, and high sensitivity to environmental changes. Its small size and short life cycle make it particularly suitable for studying physiological responses under controlled experimental conditions.
These short experiments are designed to target improvements in crop performance at the cellular level, which will directly translate into enhanced plant growth and yield. Specifically, the experiments focus on two critical organelles: the chloroplast and the mitochondrion. The chloroplast is responsible for photosynthesis, where light energy is converted into chemical energy, while the mitochondrion plays a central role in cellular respiration and energy production.
Environmental factors such as light intensity, carbon dioxide (CO₂) concentration, and nutrient availability directly influence the efficiency of these organelles. By manipulating these conditions, this experiment series aims to understand how cellular processes can be optimised to improve overall plant productivity.
Through these investigations, students will gain insights into how external environmental factors regulate internal physiological mechanisms, ultimately linking cellular function to whole-plant performance and crop yield improvement.
2. OBJECTIVES
i. To evaluate the effect of light and nutrient sources on duckweed growth and physiological responses.
ii. To determine the effect of elevated CO₂ concentration on duckweed performance.
iii. To investigate the interaction between environmental factors (light, CO₂, and nutrients) on plant physiological responses.
3. GENERAL MATERIALS AND EQUIPMENT
Duckweed (Lemna spp.)
Transparent plastic cups (150–200 mL capacity)
Paper cups (light shielding)
Ruler or imaging device
Camera or smartphone
Analytical balance
EXPERIMENT 1: THE IMPACTS OF LIGHT CONDITIONS AND NUTRIENT SOLUTIONS ON DUCKWEED GROWTH AND PHYSIOLOGICAL RESPONSES
OBJECTIVE
To determine the effect of different light quality, intensity and type of nutrient solutions on duckweed biomass accumulation and physiological responses
HYPOTHESIS
i. Duckweed cultivated under LED lights will exhibit a higher growth rate and greater biomass accumulation compared to those grown under indirect natural sunlight.
ii. Different nutrient sources will result in significantly different growth rates in duckweed due to variation in nutrient availability and form.
EXPERIMENT VARIABLES
Constant Variable (CV): Duckweed accession (Sampled from Central Perak).
Manipulated Variable (MV): Light source and Nutrient solutions
Responding Variable (RV): Growth and physiological performance
EXPERIMENT TREATMENTS
Factor 1: Type of light source
L1: LED light source
L2: Natural sunlight
Factor 2: Nutrient solutions
L1: Blood meal solution (1% wt/vol)
L2: Feather meal solution (1% wt/vol)
L3: Urea solution (1% wt/vol)
L4: Christmas Island Rock Phosphate (CIRP) solution (1% wt/vol)
L5: White sugar solution (1% wt/vol)
L6: Malacca palm sugar solution (1% wt/vol)
L7: Pond water (Control)
No. | Treatment 1: Light source | Treatment 2: Nutrient solutions | Treatment combination |
1 | LED light (L) | Blood meal (B) | L:B |
2 | Natural sunlight (N) | Feather meal (F) | L:F |
3 |
| Urea (U) | L:U |
4 |
| CIRP (C) | L:C |
5 |
| White sugar (W) | L:W |
6 |
| Malacca palm sugar (M) | L:M |
7 |
| Pond water (P) | L:P |
8 |
|
| N:B |
9 |
|
| N:F |
10 |
|
| N:U |
11 |
|
| N:C |
12 |
|
| N:W |
13 |
|
| N:M |
14 |
|
| N:P |
|
|
| “:” means combine |
MATERIALS
LED grow lights
Duckweed (Lemna sp.)
Transparent plastic cups (150–200 ml capacity)
Paper cups (light shielding)
Nutrient solutions (blood meal, feather meal, urea, CIRP, white sugar, Malacca palm sugar, pond water)
Ruler or imaging device
Camera or smartphone
Analytical balance
Light meter
Tray
EXPERIMENTAL LAYOUT & DESIGN
The experiment was arranged in a Nested Randomised Complete Block Design (NRCBD) to account for spatial and environmental variability. Two blocks were established to represent vertical environmental gradients (e.g., temperature, humidity, and light distribution).
Within each block, light condition (LED and natural sunlight) was assigned as the primary grouping factor based on fixed spatial locations. Experimental trays were nested within each light condition and block, serving as independent replicates.
Within each tray, the seven nutrient treatments were randomly assigned to individual cups, resulting in a hierarchical structure where cups were nested within trays, and trays were nested within light conditions and blocks.
This design minimises pseudo-replication by treating trays as the true experimental units for the light factor, while nutrient treatments are evaluated at the cup level within each tray.

Component | Specification | Details |
Blocks | 2 Vertical Blocks | Controls for spatial variance (e.g., temperature/humidity gradients). |
Main Factor A | Lighting (2 levels) | LED Light Source vs. Natural Sunlight. |
Main Factor B | Nutrients (7 levels) | Blood meal, Feather meal, Urea, CIRP, Pond water, White sugar, Palm sugar. |
Replicates | 3 Trays | Each Block/Light combination contains 4 independent trays. |
Total Treatments | 14 Combinations | Calculated as: 2 Light levels x 7 Nutrient levels. |
Quantitative Breakdown (n-value)
Experimental Unit: One individual cup containing Lemna.
Cups per Tray: 7 (One for each nutrient treatment).
Trays per Light Source: 8 trays (2 Blocks x 3 Replicates).
Total Experimental Units (n): 84 Cups
Calculation: 2 Light Sources x 6 Trays x 7 Cups = 84.
PROCEDURES
A. Preparation of Nutrient Solutions (1% wt/vol using Pond Water)
Collect pond water from a single source before use to ensure consistency across treatments
Prepare 1% (wt/vol) nutrient solutions (e.g. 1 g of material in 100 mL of pond water).
Prepare the following solutions:
Blood meal solution (1% wt/vol)
Feather meal solution (1% wt/vol)
Urea solution (1% wt/vol)
CIRP solution (1% wt/vol)
White sugar solution (1% wt/vol)
Malacca palm sugar solution (1% wt/vol)
Stir each solution thoroughly until evenly mixed.
Use pond water without any addition as the control treatment.
B. Preparation of Experimental Units
Label all cups according to treatment and block (e.g., B1–L:B, B2–N:U).
Nest each transparent plastic cup inside a paper cup to prevent lateral light penetration.
Add approximately 150 mL of the respective nutrient solution into each plastic cup.
C. Duckweed Inoculation
Select healthy, uniform duckweed fronds
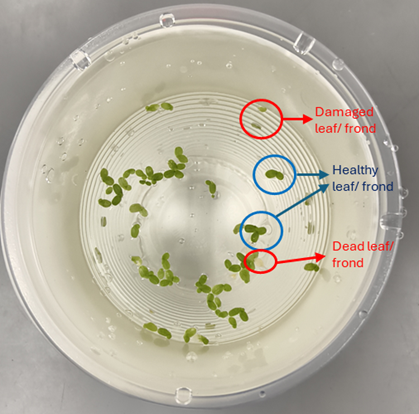
Gently rinse with clean water to remove contaminants (if necessary).
Inoculate each cup with:
3 similar-sized duckweed fronds
D. Experimental Setup (Nested Randomised Complete Block Design (NRCBD)
1. Arrange the experiment according to a Nested RCBD with two blocks:
o Blocks (2): Represent spatial/environmental gradients (e.g., temperature, humidity).
o Primary grouping factor: Light condition (LED vs. natural sunlight), assigned based
on fixed locations.
o Replicates: Three trays are nested within each light condition and block, serving as
independent experimental units.
o Nested factor: Seven nutrient treatments are applied to cups within each tray.
2. Placement of experimental units:
o LED treatments: Standardise the photoperiod (e.g., 12 hours light / 12 hours dark)
o Natural light treatments: Place trays near a window receiving indirect sunlight,
ensuring minimal exposure to direct midday light.
3. Within each tray:
o Randomly assign the seven nutrient treatments to individual cups.
o Ensure uniform spacing between cups to minimise competition and shading effects.
4. General arrangement control:
o Maintain consistent distance between trays and light sources within each light
condition.
o Avoid cross-shading and ensure similar airflow conditions across all treatments.
E. Maintenance
1. Maintain water levels by topping up with nutrient solution to the top line of the cup every two days to account for evaporation.

2. Conduct the experiment for 10 -14 days
DATA COLLECTION
Data should be recorded every 2–3 days throughout the experimental period.
A. Growth Monitoring (Non-Destructive)
At each sampling interval, record the following:
Aerial Image of Duckweed
Capture a clear top-view (aerial) photograph of each sample.
Include a scale reference (e.g., ruler or known diameter object) in every image.
Ensure consistent:
Camera height
Lighting conditions
Background (preferably plain and contrasting)
Frond Area Coverage
Analyse images using the Easy Leaf Area application to estimate surface coverage.
Record the percentage of area covered by duckweed.
·
Light Measurement (GPS Status App)
Open the GPS Status & Toolbox app.
Measure the light intensity at the level of your plants.
Record the value in Lux.
Required Tools for Image Analysis and Light reading
Easy Leaf Area App & GPS Status Apps
https://www.scenseme.com/post/3-ways-to-measure-cassava-leaf-area-which-method-is-the-best
Application guide:
https://www.scenseme.com/post/agr3301-crop-physiology-duckweed-growth-analysis-lab-guide
(Refer to video section: 5:23 – 6:55)
B. Final Day Measurements
At the end of the experiment, perform the following measurements:
Physiological Measurements
Measure photosynthetic parameters using the LI-6800 Portable Photosynthesis System.
Fresh Weight
Gently collect duckweed samples.
Remove excess surface water using tissue paper.
Weigh immediately to obtain fresh biomass (g).
Dry Weight
Dry samples in an oven at 60–70°C for 48 hours or until constant weight.
Record dry biomass (g)




Comments